Third-Party Lab Testing: COAs, Batches & What to Check
“Third-party lab tested” is one of the most common trust phrases in the supplement world.
But that phrase can mean anything — from real, verifiable batch testing to a vague marketing claim with no proof.

This guide breaks down third-party lab testing in plain English: what a COA (Certificate of Analysis) is, how batch/lot testing works, what to look for on a real report, and the red flags that suggest “lab tested” is just branding.
If you’re building your label-reading skills, start with our foundational guide to reading supplement labels and spotting fairy dusting.
For another common phrase that gets misunderstood, see what “clinically studied ingredients” really means on supplement labels.
Last updated: April 2026
Written by Daniel Popa, founder of LiveGoodForLife.com. This content is for educational purposes only and is not medical advice.
What Is Third-Party Lab Testing?
Third-party lab testing means a supplement is tested by an independent laboratory (not the brand’s own internal team). The goal is simple: verify that the product matches what the label implies — at least in basic ways like identity, purity, and (sometimes) potency.
It’s important to keep expectations realistic. Third-party testing does not automatically prove a supplement “works.” It usually verifies things like:
- Identity: the ingredient is what it claims to be
- Purity: checks for common contaminants (varies by product type)
- Potency (sometimes): active compounds match a stated target
The NIH Office of Dietary Supplements explains that supplements are regulated differently than drugs and are not required to prove effectiveness before being sold. That consumer context matters when interpreting “tested” language on labels. See the NIH overview on dietary supplements for consumers.
COA Explained: What a Certificate of Analysis Actually Is
A COA (Certificate of Analysis) is a document that shows test results for a specific product (or ingredient) and ideally a specific batch/lot.
Think of it like a receipt for testing: it’s not a guarantee of results — but it’s evidence that testing happened and that results were recorded.
A COA is only valid if it matches your exact batch
This is the detail most people miss. A COA that isn’t tied to a lot number (or batch ID) is much less meaningful, because you can’t confirm it applies to the bottle you’re holding.
Batch and Lot Numbers: The Missing Link in Most “Lab Tested” Claims
Batch/lot testing means the company tests a specific production run. That run gets a lot number, and the COA should reference it.
If a brand says “lab tested” but cannot provide a COA for a recent lot, you’re being asked to trust a claim you can’t verify.
This is one reason independent label reviews matter. For real-world examples of mismatches between claims and what testing found, see our summaries of the Amazon supplement label accuracy study and the weight-loss supplement label accuracy research.
What a Real COA Should Include
COAs vary by product type, but strong COAs usually include several basics that make them checkable.
| COA Item | Why It Matters | What to Look For |
|---|---|---|
| Must-have Product name + format | Prevents generic reports being reused | Exact product name (capsule/powder), sometimes SKU |
| Must-have Lot/batch number | Links the COA to your bottle | Lot ID on the COA that matches the bottle label |
| Must-have Test date + report date | Shows recency and relevance | Recent dates for active products still being sold |
| Helpful Lab name + location | Verifies a real lab exists | Named lab with contact or identifier |
| Helpful Methods / standards | Shows how results were generated | Methods listed (even briefly), not just “PASS” |
| Varies Potency / assay results | Helps confirm label meaning | Numerical results (not only checkmarks), especially for actives |
The Big Red Flags: When “Lab Tested” Is Probably Just Marketing
- No COA available (or the brand refuses to provide one)
- COA has no lot number (can’t match it to your bottle)
- Generic “PASS/FAIL” only with no numeric results or methods
- No lab name or the lab can’t be verified
- Old reports reused for years with no batch context
- COA doesn’t match the product (wrong form, flavor, serving size, or label)
These red flags often appear alongside other “trust language” that sounds scientific but doesn’t provide proof. If you see heavy claims, blends, and vague testing in the same product, revisit how fairy dusting works on labels to understand why complexity can be used as camouflage.

A Simple Checklist: How to Verify Third-Party Testing in 2 Minutes
- Find the lot number on your bottle (often near the seal, bottom, or side).
- Check if the brand provides a COA publicly or on request.
- Match the lot number on the COA to the bottle.
- Check dates (recent batches should have recent reports).
- Look for substance: named lab, methods, numeric results where relevant.
- Confirm the COA matches the product (same form, same item, same specs).
If you can’t connect the COA to your bottle, treat “third-party tested” as a nice claim, not a verified trust signal.

Comparison Table: Real Testing vs Testing-Sounding Language
| What Brands Say | What It Might Mean | What You Should Ask |
|---|---|---|
|
Common “Lab tested” | Could be real testing, or just a broad claim with no document you can match to your bottle. | Is there a COA tied to a batch/lot number that matches what I bought? |
|
Weak “Tested for purity” (no proof) | Sounds reassuring, but offers no verification if the lab, methods, and results aren’t shown. | Which lab? Which contaminants? What methods? Can I see the actual report? |
|
Stronger “COA available by lot number” | Verifiable documentation exists and can be linked to a specific production run. | Does the COA match my exact bottle lot and include dates + lab details? |
|
Best “Batch testing + clear label dosing” | Transparency supports trust: you can verify quality signals and evaluate whether dosing makes sense. | Can I verify identity/purity and also check that the formula is plausibly dosed? |
Pros & Cons of Relying on Third-Party Testing
Pros
- Improves trust when reports are verifiable and batch-specific
- Helps confirm identity and basic quality control
- Reduces “trust-me” marketing when documentation is transparent
Cons
- Testing language can be used as marketing without proof
- A COA doesn’t automatically prove effectiveness for your goal
- Not all COAs test the same things (results depend on what was tested)
Scientific & Medical Context
Research and consumer guidance repeatedly highlight that supplement labels can be misunderstood, and product quality varies. The NIH Office of Dietary Supplements provides consumer guidance on what labels can and can’t guarantee in its overview of dietary supplements.
For an example of how supplement labeling and claims are studied in real markets, PubMed indexes research examining label accuracy and consumer interpretation. One example is this PubMed record on supplement label accuracy and consumer perception.
For label-reading fundamentals (serving size, forms, and what to scan first), Healthline’s guide on how to read supplement labels is a helpful reference.
Personal Insight
When I review supplement labels for LiveGoodForLife, I treat “tested” language as a claim that must be connected to something verifiable. The simplest standard is: can this report be matched to a real batch/lot number? If not, it may still be a decent product — but the trust signal is weak.
My goal is not to create fear. It’s to help readers avoid buying “trust phrases” instead of clarity. That’s why this post connects COAs to practical label evaluation — the same approach we use across our supplement accuracy series and our LiveGood Supplement Reviews section.
LiveGood Example: How to Use a COA the Right Way (Without Guessing)
A lot of brands say “third-party tested,” but the only way to know what that really means is to check whether you can verify a Certificate of Analysis (COA) for your exact batch.
If you want a real-world example of what this should look like, use this LiveGood resource as a reference: see how LiveGood publishes product Certificates of Analysis (COAs).
The simple COA verification checklist
- Find the lot/batch number on your bottle or pouch (this is your “identity tag”).
- Match the product name and lot number on the COA to your bottle (if they don’t match, it’s not truly batch-specific).
- Look for what was actually tested (microbial, heavy metals, identity, purity—this varies by product type).
- Check the results format:
- “Conforms/Pass” is fine when the COA clearly shows the specification it must meet.
- Numeric results are even better because you can compare result vs spec at a glance.
- Watch for “label claim / by input” wording on potency lines. Sometimes a COA includes manufacturing targets rather than an independent potency assay—so treat it as a transparency signal, not automatic proof of potency.

What a strong COA typically includes (and what it tells you)
| COA Section | What to Look For | What It Helps You Confirm |
|---|---|---|
| Must-match Product + Lot/Batch ID | Exact product name and a lot number that matches your bottle/pouch | You’re reading a COA for your batch—not a generic PDF |
| Quality Microbial testing | Targets for total plate count, yeast/mold, and pathogen checks (e.g., E. coli / Salmonella) | Basic safety and cleanliness controls for powders/capsules |
| Purity Heavy metals / impurities | Clear limits (specs) and results that meet them (often shown as “conforms” or numeric) | Whether the batch meets contaminant thresholds |
| Identity Methods listed | Recognizable test methods (USP / AOAC / GC-MS depending on product) | The COA isn’t just a statement—it’s tied to specific testing procedures |
| Context Potency lines (when present) | Prefer measured assay results; if you see wording like “by input,” treat it as label-claim documentation | Helps you separate measured testing from manufacturing targets |
Quick reality check: A COA doesn’t “prove a supplement works.” What it can do is prove the batch meets key quality controls (identity/purity/microbials) and that the brand is willing to show documentation instead of asking for blind trust.
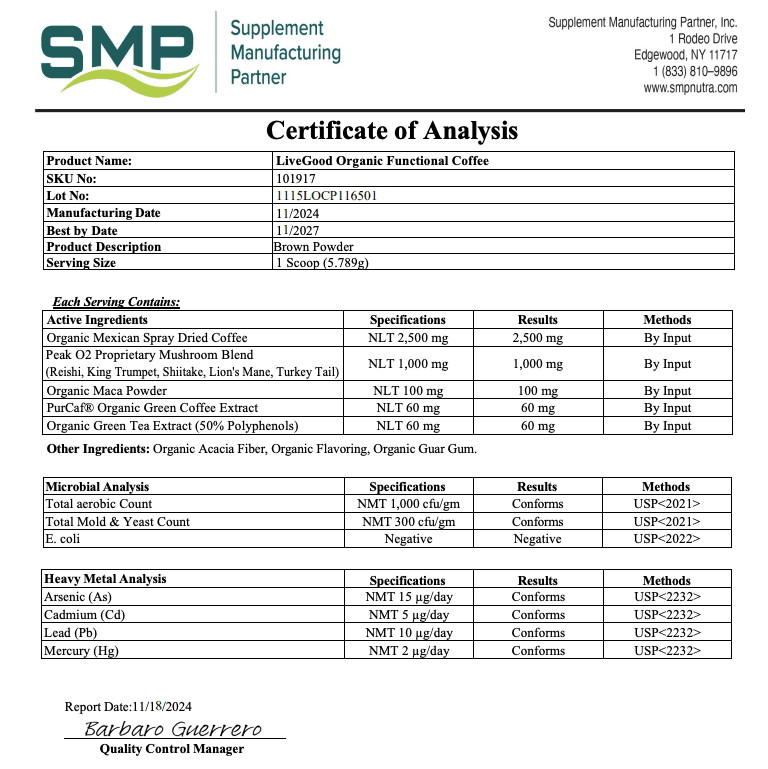

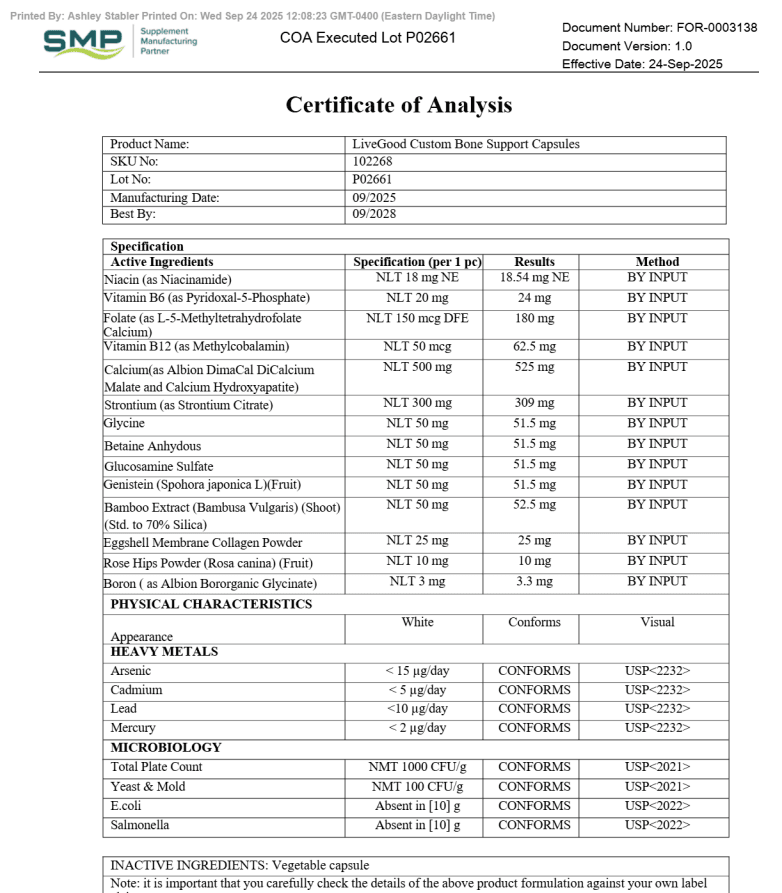
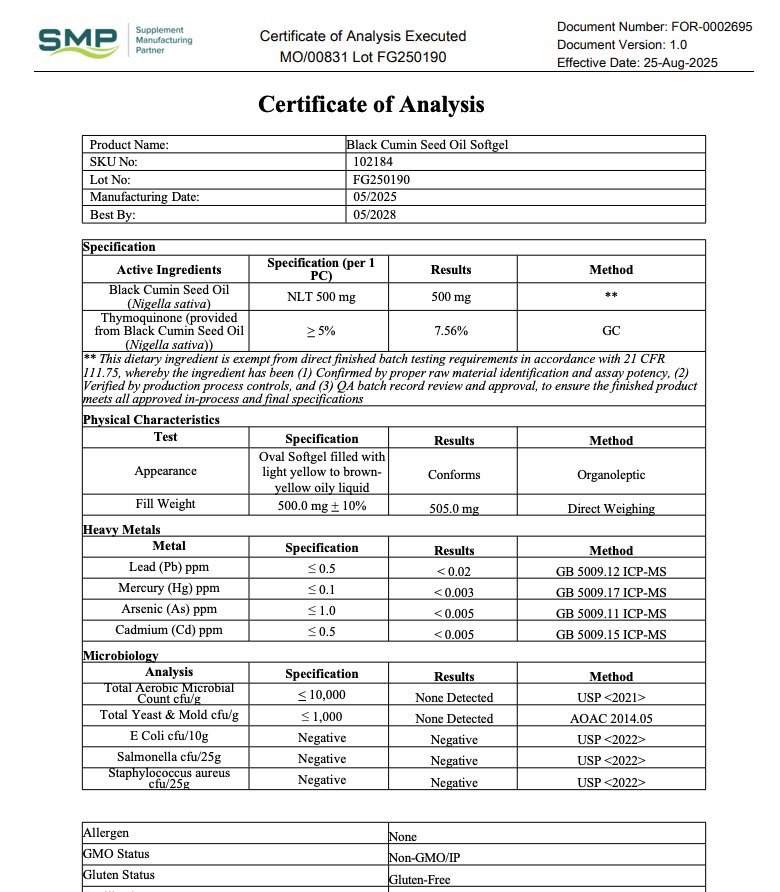
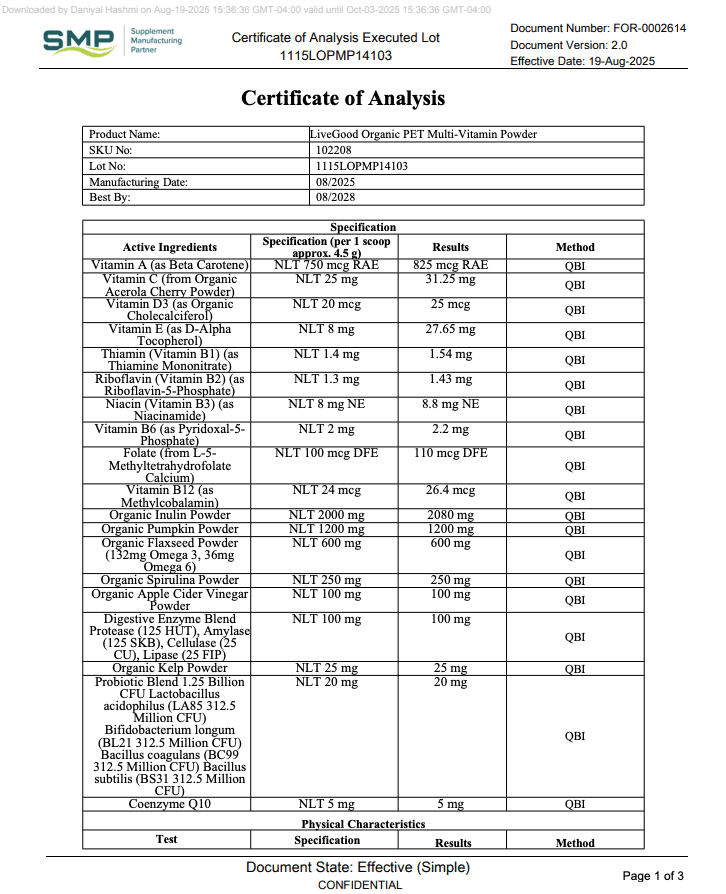
If you want a practical model to copy, use this page as your benchmark: LiveGood’s Certificates of Analysis resource.
Note for essential oils: For oils, the strongest “third-party testing” signal is often a GC-MS profile showing the chemical composition (because essential oils are complex mixtures). When a brand can show that kind of report, it’s a stronger signal of identity than vague “tested” wording alone.

FAQ: Third-Party Lab Testing and COAs
What does third-party tested mean on supplements?
It means an independent lab (not the brand) performed testing. The claim is only meaningful when the brand can provide a COA tied to a specific batch/lot number.
What is a COA (Certificate of Analysis)?
A COA is a report showing test results for a product or ingredient. Strong COAs include the product name, lab details, test dates, and a lot number that matches your bottle.
Where do I find the lot number on a supplement bottle?
It’s usually printed near the bottom, side label, or close to the seal. If there’s no lot number, it’s harder to verify any batch-specific testing.
Does a COA prove a supplement works?
No. A COA helps verify identity/purity (and sometimes potency). Effectiveness still depends on dose, form, and whether the formula matches your goal.
What are the biggest COA red flags?
No lot number, no lab name, overly generic “PASS” results, very old reports, or a COA that doesn’t match the exact product you purchased.
Is “GMP” the same as third-party testing?
No. GMP refers to manufacturing practices. Third-party testing refers to independent verification. A strong trust profile often includes both, plus transparent label dosing.
Final Thoughts
Third-party testing is one of the best trust signals in supplements — when it’s verifiable. The simplest standard is still the best: a COA that matches your exact lot number, with real lab details and meaningful information.
If you want to build a stronger label-reading system over time, keep your focus on what can be checked: transparent dosing, sensible formulas, and documentation that connects to the bottle you’re actually taking.
Disclaimer: This article is for educational purposes only and does not provide medical advice. Always consult a qualified healthcare professional for personal guidance. This post may contain affiliate links.
What If You Could Unlock the Secrets of Your Health?
Have you ever wondered what your body really needs to thrive? Imagine if you could uncover the exact steps to transform your health—effortlessly. The secret? It’s right here, and it’s free.
With the Free Health Assessment, you can finally get the answers you’ve been seeking. Whether you want to lose weight, boost your energy, or simply feel better every day, this personalized health report will point you in the right direction.
Why Take the Health Assessment?
- Personalized Health Insights: Get a deep dive into your current health, covering heart health, immune strength, stress levels, and more.
- Discover Your Hidden Potential: Uncover health risks you may not even be aware of and learn how to tackle them effectively.
- Actionable Expert Advice: Receive simple, customized tips that fit your lifestyle—whether you want to build muscle, lose weight, or improve your well-being overall.
- Quick & Easy: It only takes a few minutes, but the impact could last a lifetime.
- Absolutely Free: No hidden fees, no catch. Just a commitment to your better health.
Take the Quiz, Unlock Your Potential
Curious about what your body needs? Don’t let uncertainty hold you back any longer. Thousands have already taken their first step towards better health; now it’s your turn. This isn’t just a questionnaire—it’s your gateway to a healthier, more vibrant life.
Start Your Free Health Assessment Now







